Designing Dual Catalyst for Hydroamination:
We are currently working on assembling heterometals into a molecular
catalytic system so that both metals can act catalytically for two
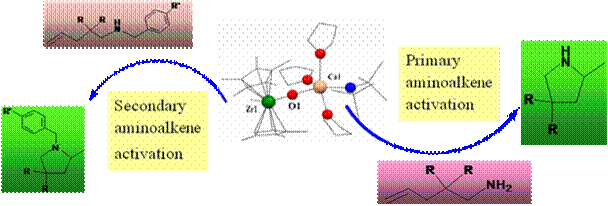
different reactions. For example, we have recently shown that Ca and Zr metal centers can be assembled resulting a heterobimetallic
catalyst which functions as dual catalyst in intramolecular
hydroamination reactions of primary amino alkenes and secondary
amino alkens. The Ca center cyclizes the primary aminoalkenes and
the zirconium center cyclizes the secondary aminoalkenes.
Mukherjee et al. Angew.
Chem. Int. Ed.
2011,
50, 3968-3972 and
RSC Adv.,
2013, 3, 1255–1264.
Phenalenyl Non-Bonding Orbital for Organometallic Catalyst Design:
We are working on the development of molecular catalyst utilizing
the nonbonding orbital of phenalenyl ligand system.
Phenalenyl is a well-known odd alternant hydrocarbon with high
symmetry (D3h) which has the ability to
form three redox species: cation, neutral radical, and anion.
It has been demonstrated by Haddon and co-workers that the lowest
unoccupied molecular orbital (LUMO) of spirobiphenalenyl boron
derivatives posing cationic state of phenalenyl can readily accept
one electron to become open shell molecular system
leading to the development of the best known neutral organic
conductor.
The unpaired electron of neutral radicals serve as the charge
carriers and orbital overlap between adjacent radicals generates the
conduction pathway through the lattice in solid state. In this way a
new genesis of multifunctional electronic and magnetic materials
exhibiting simultaneous bi-stability in three physical channels:
magnetic, electrical, and optical and earlier elusive resonating
valence bond (RVB) ground state in the solid state have been
realized through open shell phenalenyl based molecular system.
We postulated that the vacant nonbonding orbital of cationic
phenalenyl can not only be used in building of molecular conductors
by electron acceptance but also it may have a great influence in
catalytic reactions. The metal complexes of the phenalenyl based
ligand systems are expected to behave as highly Lewis acidic. Very
recently we have shown that the cationic state of phenalenyl based
molecular system can be used for development of molecular catalyst
for ring opening polymerization of cyclic esters which works
through electron acceptance into the LUMO of organoaluminum
phenalenyl derivatives bearing the cationic state of phenalenyl
moiety. In a recent study we demonstrated for the first time that
the empty orbital of phenalenyl moiety can be used to tune the
catalytic activity in ring opening polymerization reaction.
Sen et al.
Chem. Eur. J. 2012,
18, 54-58.
Mukherjee et al.
Chem. Eur. J.
2012, 18,
10530-10545
Sen et al.
Dalton Trans.
2013,
42,
1893-1904.
Spin
Selective Organometallic Zinc Phenalenyl
Complex:
Interaction between a ferromagnet and an organic molecule provides a
complex study of interface chemistry and magnetism. However, the
possibility to engineer these interface effects, by virtue of
controlling the interface parameters such as molecular structure,
morphology and electronic property of interface holds promise in
realization of novel functional molecular spintronic devices.
Magnetoresistance, defined as the change in resistance with applied
magnetic field, in molecular devices such as vertical organic
spin-valves and magnetic tunnel junctions involves two magnetic
electrodes with spin diffusive or tunneling transport through the
organic spacer layer. In a recent ongoing, we demonstrate a new
interface phenomenon of a spin-filter effect using a single
ferromagnet electrode and a phenalenyl derivative which may be
considered as a mini fragment of a graphene sheet resulting in a
large magnetoresistance of more than 20% close to room temperature.
The existence of nonbonding orbital of the phenalenyl molecule leads
to a open shell radical formation on interaction with the FM layer
plays a key role in this interface phenomena.
This findings including the
possibility to use π-conjugated
organic molecular systems as interface templates to selectively
engineer the interface mechanisms will open up rich prospects for
fundamental study and development of future molecular spintronic
devices.
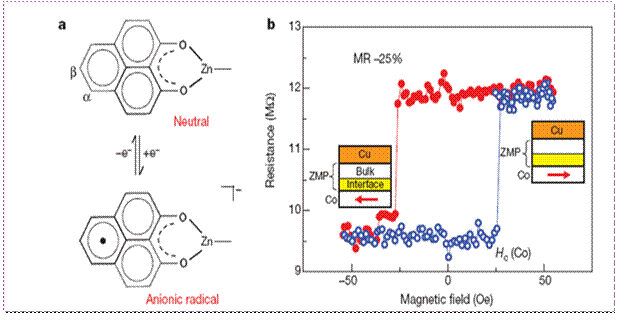
The
first organometallic molecular based memory device that works
at near room temperature.
(Raman et al.,
Nature, 2013,
493, 509-513)
Abnormal N-Heterocyclic Carbene: Excellent Building Block to Design
Catalyst:
Nearly two decades ago the isolation of heteroatom stabilized
carbenes [normal N-heterocyclic carbenes (nNHCs)] by Arduengo
has led to numerous breakthroughs in organometallic and organo-catalysis.
In 2009, Bertrand and co-workers isolated a new class of carbene in
which the carbene center is no longer located between the two
nitrogen atoms but is generated between nitrogen and carbon atoms.
This new class of carbenes is known as abnormal NHCs (aNHCs).
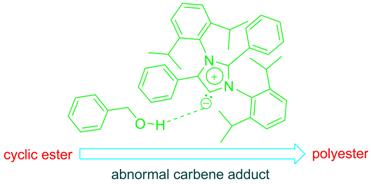
We recently established the first use of aNHC, in
organocatalytic ring opening polymerization of three different
cyclic esters rac-lactide (rac-LA), ε-caprolactone
(ε-CL) and δ-valerolactone (δ-VL) will be
discussed.3The catalytic results unravel that 1 is
efficient organocatalyst for ring opening polymerization at ambient
temperature. This aNHC exhibited the fastest catalytic
activity for ring opening polymerization of ε-caprolactone
among any NHCs reported till date.
Sen et al.
Chem. Commun.
2011, 47,
11972–11974.
Recently in another study, we accomplished the synthesis of
halobridged palladium dimers containing abnormal NHC for Suzuki–Miyaura
cross coupling of aryl chlorides. Palladium complexes bearing
abnormal N-heterocyclic carbene were used as catalysts in Suzuki–Miyaura
cross coupling of aryl chlorides at room temperature. The catalyst
remained active for 10 successive catalytic runs and can activate
4-chlorotoluene at ambient temperature with 0.01 mol% catalyst
loading resulting in a TON of 9500 within 6 h.
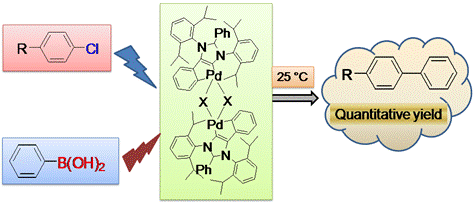
Sau
et al. Chem. Commun.
2012,
48, 555-557.
Supported Palladium Nanocatalyst as Recyclable Catalyst for C-C
Coupling:
We are actively working on development of hetergenous palladium
nanocatlyst for C-C coupling reactions which can be recycled.
Recently we established a convenient method to develop palladium
nanoparticles (PdNPs) embedded into a polymer matrix, PPS [PPS =
poly(1,4-phenylene sulphide)] as well as with chemically modified
single walled carbon nanotubes via the thermolysis of
palladium acetate. These palladium nanoparticles (PdNPs-PPS) have
been used as an efficient heterogeneous nanocatalytic system for
copper free acyl Sonogashira reaction. A wide range of ynones was
synthesized in high yields under mild reaction condition.
The catalyst was recovered and recycled up to five times.
Transmission electron microscopy (TEM) images revealed that the
catalyst maintained nanospheric dimensions till four consecutive
catalytic cycles. This simple protocol was further explored in
one-pot multicomponent synthesis of 2,4-disubstituted pyrimidines
and a tetrahydro-β-carboline derivative in improved yield.
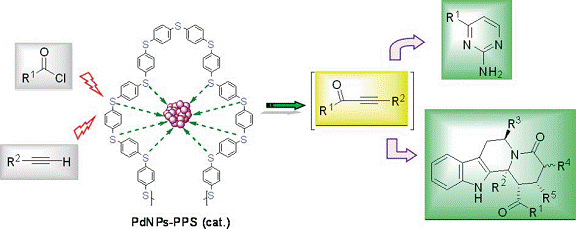
Santra
et al.
Green Chem.
2011,
13, 3238-3247 and
RSC Adv.
2012,
2,
7523–7533